 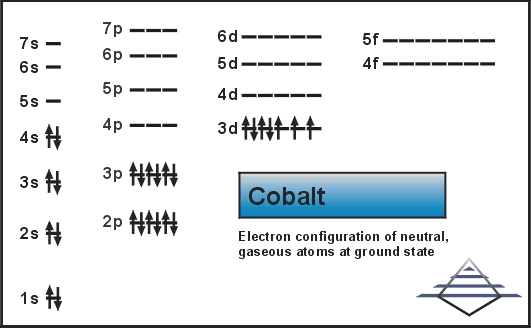
This is the broader view of the electron configuration for the element which discloses all the aspects of the element.įor instance, you can study and understand the distribution of electrons of this element in its shells. We here advise them all to go through the exclusively available full-fledged electron configuration of the element. We believe it will ultimately be worthy for all those enthusiasts who are willing to have a thorough understanding of Cobalt.Īs we have already discussed the significance of the electron configuration of Cobalt to all our readers. You can refer to and study the electron configuration of the element in order to develop a thorough understanding of the same. You can best understand the whole distribution process of the ions by understanding their electron configuration. Due to the same charge these atoms may either gain or lose the electrons during the electron configuration process. The ion of Cobalt is basically those atoms that directly bear the impact of charge upon them. You can also read or study the same in the periodic table of elements to find the Cobalt presence there. The electron configuration seems like an equation that gives a specific identity to the chemical element. This whole process takes place in the atomic orbital of the element and gives the most simplified form to it. 
The electron configuration in the easy or layman language is the process in which the element basically makes the distribution of its electrons to its subshells. As a scholar or even a chemist, you will most probably come across the electron configuration part of the element during your study and research. The electron configuration of any chemical element is its most significant chemical property that one should always be aware of. These batteries are defining and ruling the arena of modern-day futuristic e vehicles. The best modern-day usage of Cobalt is in the production of lithium-ion batteries. In the commercial domain, the application of Cobalt is quite expensive. The maximum quantity of Cobalt is today extracted as the by-product of copper and also from the mining of nickels. Today the production of Cobalt is possible in several forms and the element is available in significant quantity accordingly. There is evidence that states the usage of this element as early as in the period of 1300 or so. The history of the Cobalt chemical element is quite rich in itself as it is one of the oldest chemical elements in the world. Furthermore, the element belongs to the d block of group 9 in the periodic table. It has the general appearance of a hard silvery and lustrous metal just like any pure metal object. 
It’s much like the Nickel that you can extract from the crust of the earth in the form of combined chemical elements. The element is available for the whole lot quantity in the crust of the earth. In Chemistry or biology Cobalt is a name of a chemical element that comes with its atomic number 27 and the symbol of Co. As we progress ahead with the article the readers will find the different chemical aspects of Cobalt that they can read for their learning of the element. We believe if you are someone who is willing to explore this chemical element well for their academics and other requirements then this article will definitely come in handy for themselves. The article ahead contains some significant information on the electron configuration, valence electrons, and the other significant chemical properties of the element. Tin mining began in Australia in 1872, and today tin is used extensively in industry and commerce.Study the Cobalt Electron Configuration and get to know this chemical element from a closure perspective. Tin shares chemical similarities with germanium and lead. Bending a bar of tin produces a characteristic squealing sound called "tin cry". It forms a number of useful low-melting alloys (solders) which are used to connect electrical circuits. It has been used for many years in the coating of steel cans for food because it is more resistant to corrosion than iron. It has the look of freshly cut aluminum, but the feel of lead. The metal is silvery white and very soft when pure. Named after the Etruscan god Tinia, the chemical symbol for tin is taken from the Latin stannum. Early metal smiths were quick to learn that mixing copper with tin created a more durable metal (bronze), and it is principally for its alloys that tin is valued today. Mentioned in the Hebrew scriptures, tin is of ancient origins. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
